
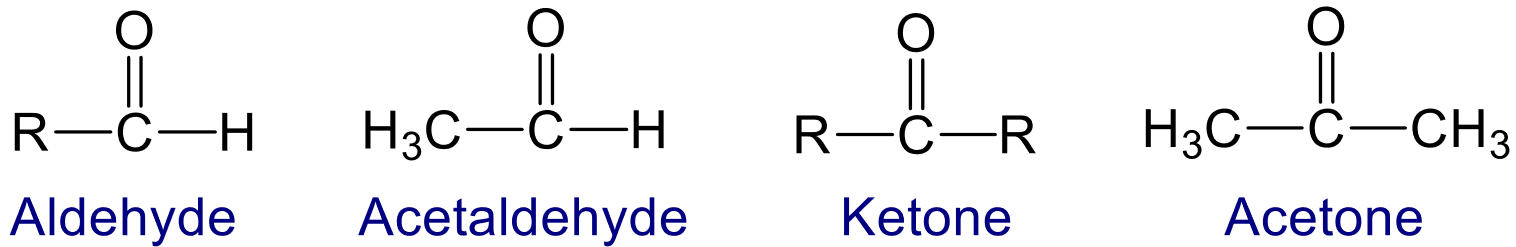
Aldehyde and Ketone
Class 12 Chemistry Aldehydes and Ketones NEB Notes 2080. Class 12 Chemistry Unit 13 Aldehydes & Ketones Complete note, Exercise, Important Questions.
Introduction
Aldehydes and ketones are commonly called as carbonyl compound because they contains carbonyl (>C=0) functional group. This carbonyl group largely determines the chemical properties of aldehydes and ketones.
They are represented by general molecular formula CnH2nO.

Aldehyde exist in the form of –CHO functional group which is formyl group and ketone exist in the
form of >C=O functional group which is keto or oxo group.
Structure of aldehyde and ketones

Classification of aldehydes and ketones
- Aliphatic aldehyde and ketone
- Aromatic aldehyde and ketone
Aliphatic aldehyde and ketones

In aliphatic aldehyde and ketone, the carbonyl carbon is bonded to alkyl group except formaldehyde.
Nomenclature of aldehydes and ketones
General formula:
Prefix + word root + primary suffix + secondary suffix
- Prefix: side chain (branch) substituent
- word root: number of C – atoms
- primary suffix: nature of C- atom
- secondary suffix: functional group ( -CHO ⟶ al, >C=O ⟶ one)

Isomerism in aldehyde and ketone
1. Chain isomerism
Aldehydes and ketones having same molecular formula but different number of carbon chain length are called chain isomers. C4H8O.
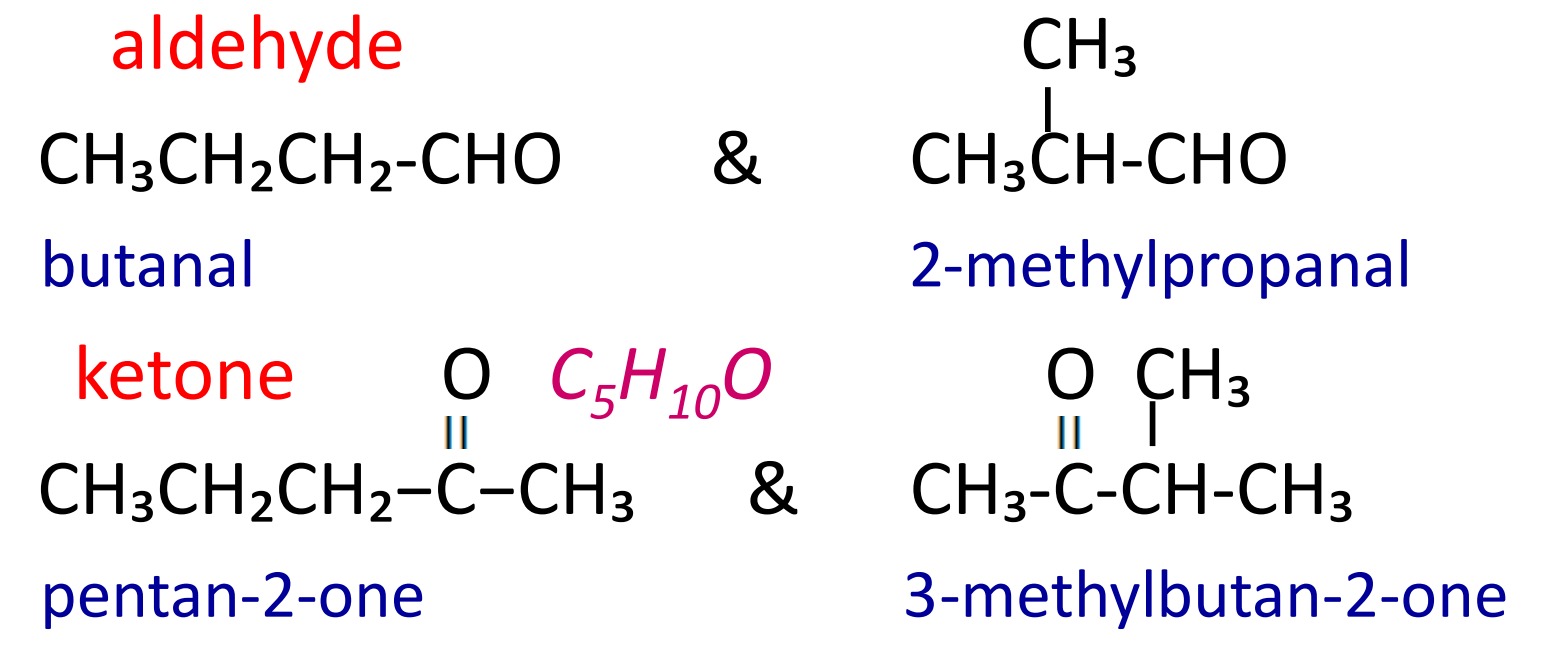
2. Position isomerism
Ketones having same molecular formula but different position of >C=O group on carbon chain are called position isomers. (-CHO group always gets position 1)

3. Functional isomerism
Aldehydes and ketones having same molecular formula but different functional group are called functional isomers.

4. Metamerism
Ketones having same molecular formula but different number of carbon atoms on either side of >C=O functional group are called metamers. This phenomenon is called metamerism.

5. Tautomerism
The isomerism which arises due to inter-convertible hydrogen atom are called (keto-enol) tautomerism.

General methods of preparation of aldehydes and ketones
1. Oxidation of alcohol
Aldehydes and ketones are prepared by the oxidation of primary and secondary alcohols by using different oxidizing agent KMnO4 , K2CrO7 ,PCC etc in acidic medium.

*Pyridium chlorochromate(PCC) can be used
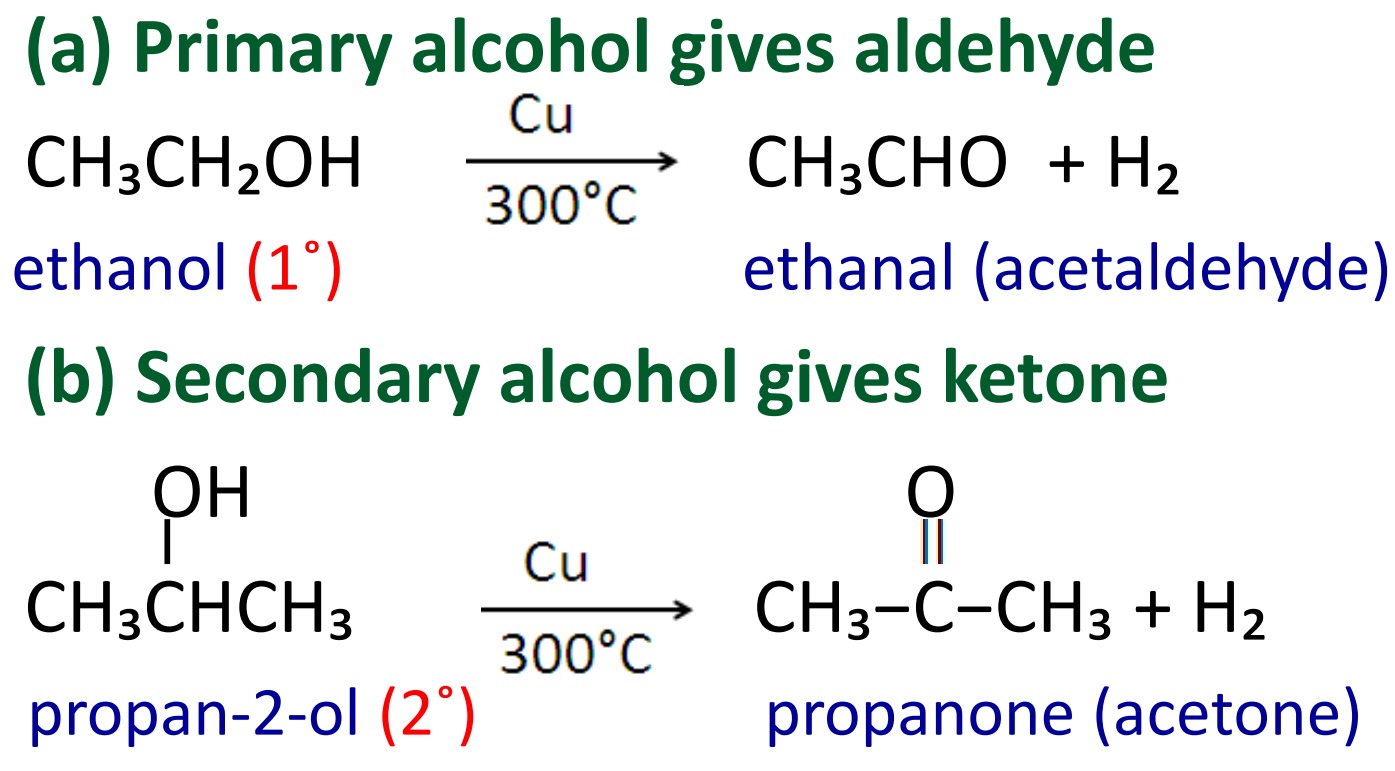
2. Catalytic dehydrogenation of alcohol
Aldehydes and ketones are prepared by heating primary and secondary alcohols with copper metal at 300˚C.
3. Ozonolysis of alkenes
When alkene reacts with ozone in presence of CCl₄ as inert solvent then ozonoid is formed which further hydrolysed with zinc water then two molecules of carbonyl compounds (aldehydes and ketones) are formed.

Task: Q. Prepare acetaldehyde and acetone by using ozonolysis reaction.
4. Reduction of acid chloride (Rosenmund reaction)
Aldehydes are prepared by the reduction of acid chloride (acyl chloride) with hydrogen in presence of Pd and BaSO₄ poisoned by quinoline.
.jpeg)
Ketones are prepared by heating acid chloride with dialkyl cadmium.

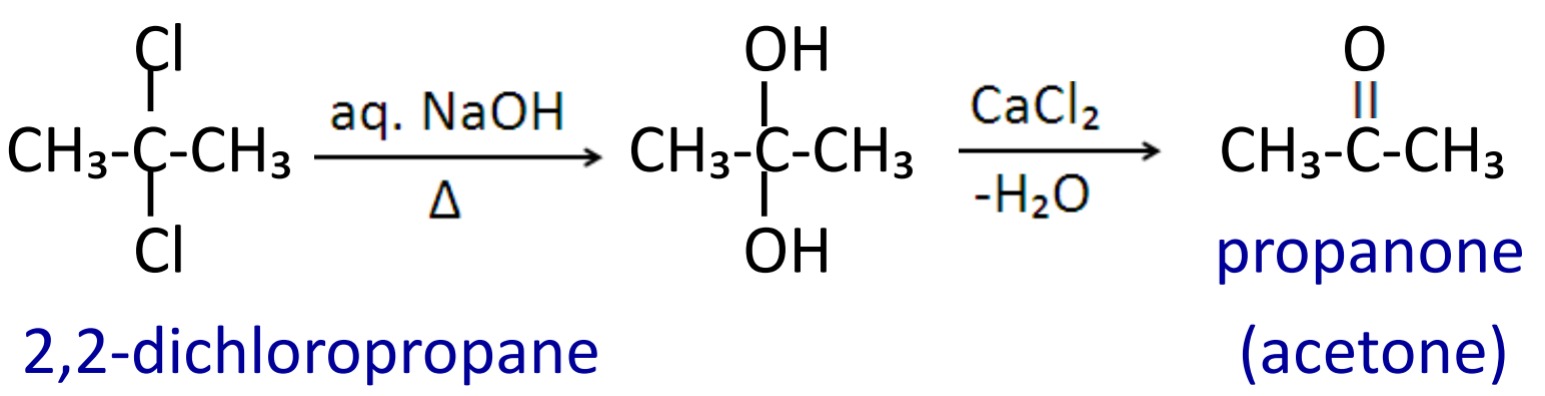
5. Hydrolysis of geminal dihalide
When geminal dihalides are heated with aqueous NaOH or KOH followed by dehydration with anhydrous CaCl₂ then aldehyde are formed.

Similarly ketone can be prepared by dihalides which are bonded to non terminal carbon atom.
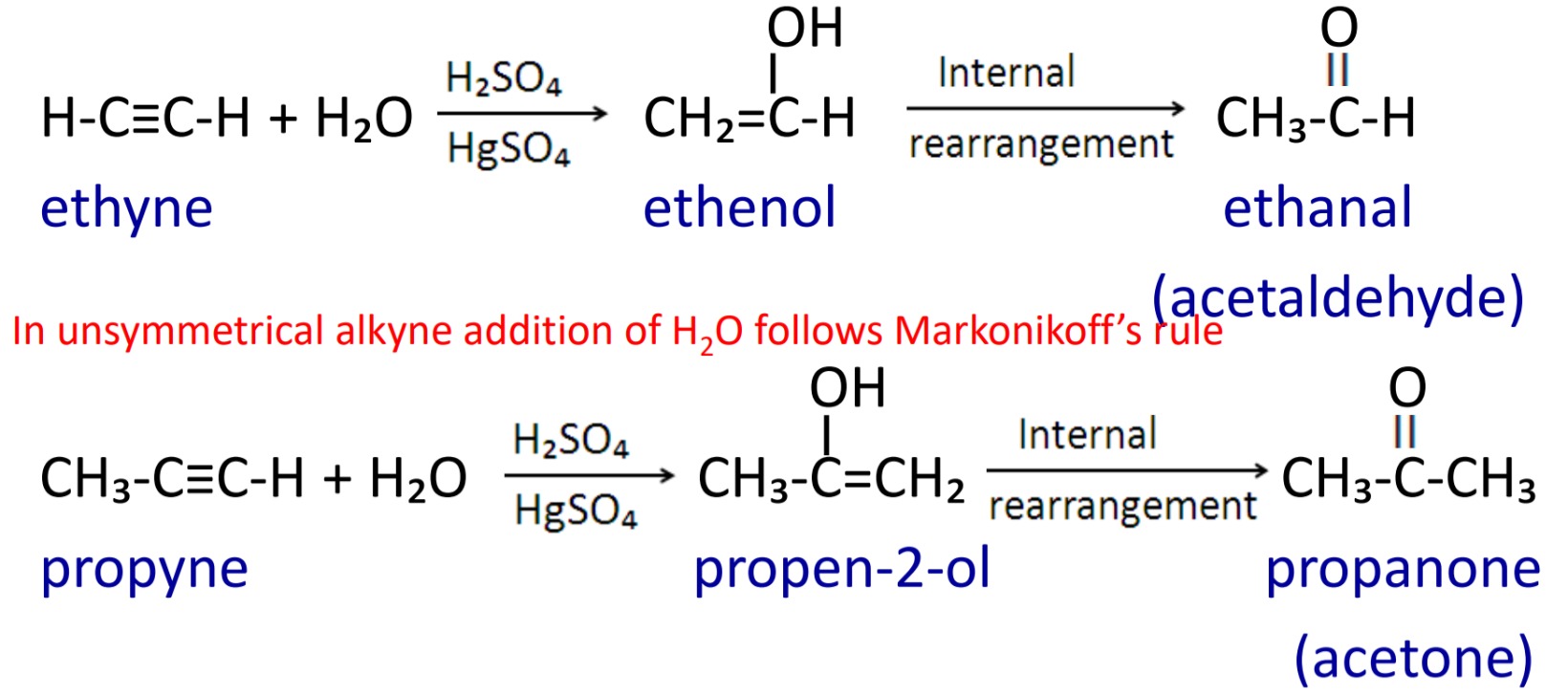
6. Catalytic hydration of alkynes
When alkyne reacts with water in presence of dilute H₂SO₄ and HgSO₄ as catalyst at about 60˚C then alkenol is formed which undergoes internal rearrangement (Tautomerism) then aldehyde and ketones are formed.
Physical properties of aldehydes and ketones
1. Formaldehyde is gas. Other aldehyde and ketone up to C-11 are colorless liquids and higher are colorless solids.
2. Lower aldehyde possess unpleasant smell and higher aldehyde and lower ketones have sweet pleasant odour.
3. Lower aldehyde and ketones (C-5) are soluble in water, higher are partially soluble and completely soluble in almost all organic solvents like benzene, ether, alcohols, etc.
4. They have higher boiling point than alkanes and lower than that of alcohols and carboxylic acid having comparable molecular mass.
Chemical properties of aldehydes and ketones
[A] Nucleophilic addition reaction.
The carbonyl group of aldehyde and ketone is partially polarized due to difference in electro negativity value between carbonyl carbon and oxygen atom. Thus positively charged carbon is easily attacked by a nucleophile and gives the addition product.

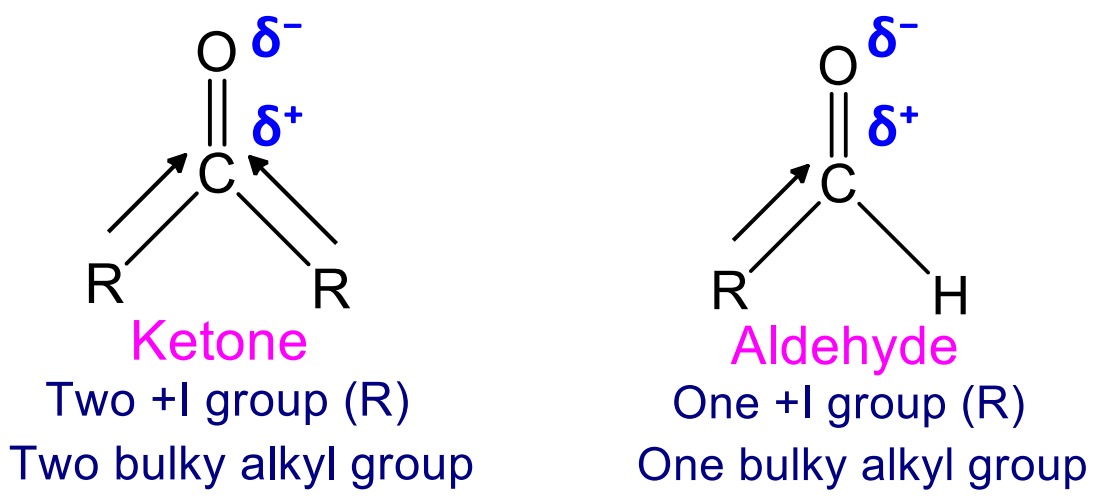
Aldehydes are more reactive than ketone towards nucleophilic addition reaction because,
1. In ketone, carbonyl carbon is bonded to electron releasing two alkyl groups which makes carbonyl carbon less electropositive and less reactive toward nucleophilic attack but aldehyde is bonded to one alkyl group.
2. Similarly ketone consist of two bulky alkyl groups which cause steric hinderance for attack by nucleophile but aldehyde consist of only one alkyl group and attack of nucleophile is easy.
1. Addition of hydrogen (Catalytic hydrogenation)
When aldehydes and ketones are reduced with hydrogen in presence of Ni or Pt catalyst then alcohols are formed.
.jpeg)
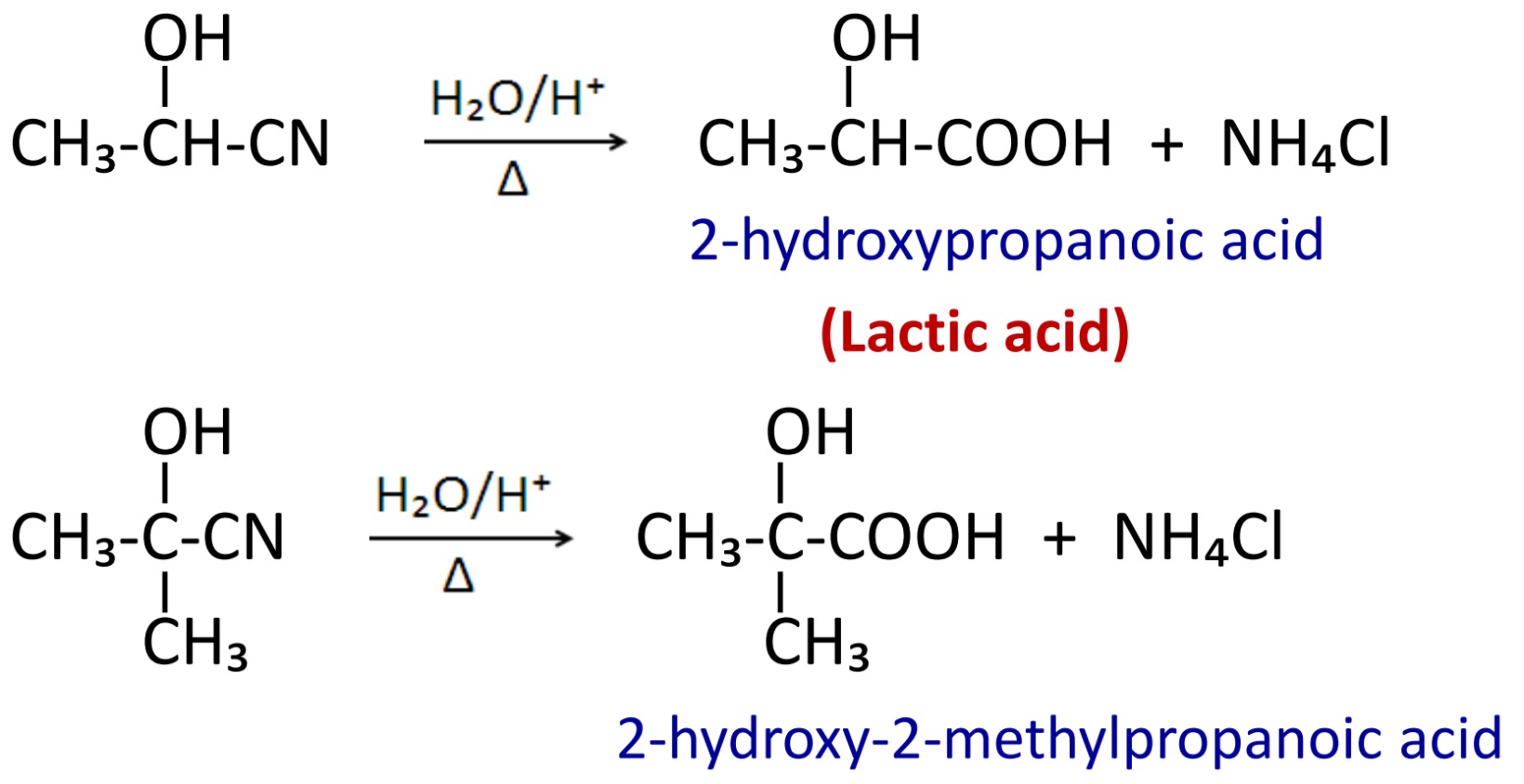
h52. Addition of hydrogen cyanide (HCN)
When aldehyde and ketone reacts with HCN in acidic medium then cyanohydrin are formed.
.jpeg)
Like other nitrile, cyanohydrin can be hydrolysed in acidic medium to give α-hydroxy carboxylic acid.
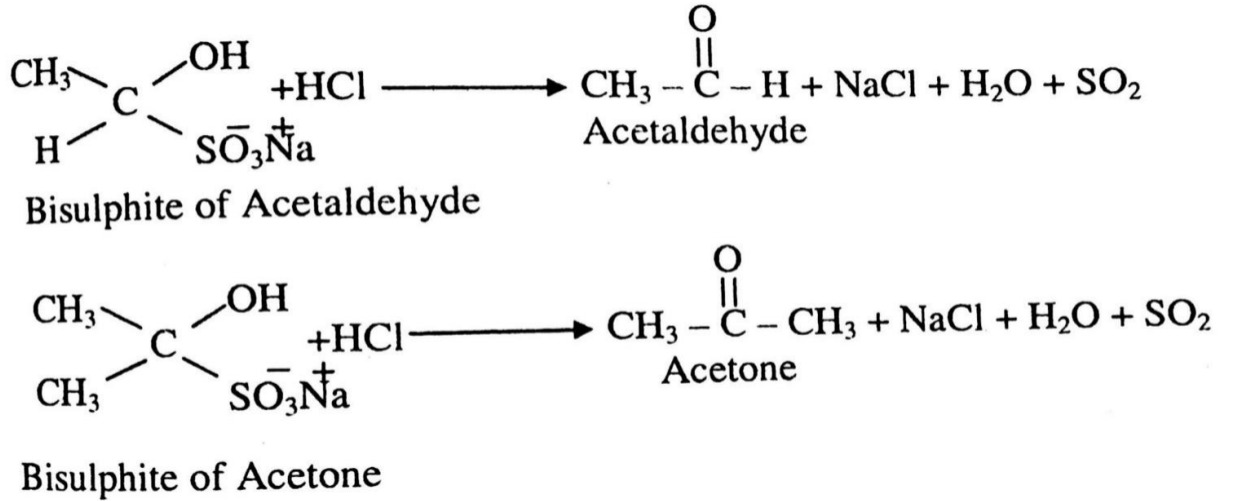
3. Addition of sodium bisulphite
When aldehyde and ketone reacts with sodium bisulfite then crystalline solid of addition product is formed.
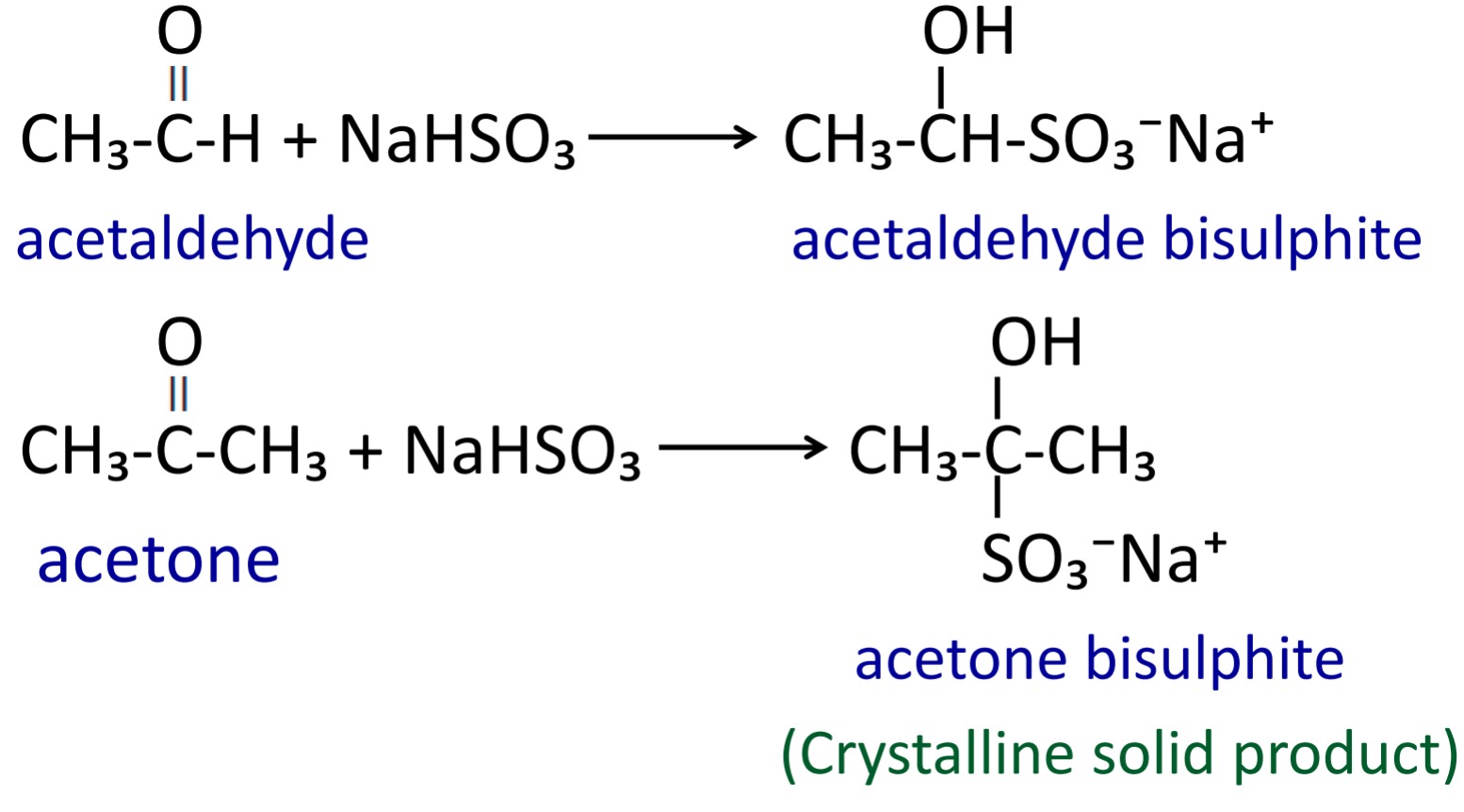
These bisulphites further react with mineral acid or alkali, we get original aldehyde & ketone.
[B] Addition reaction followed by elimination
Aldehyde and ketone add to ammonia derivatives followed by removal of water molecule. (Formation of >C=N bond)
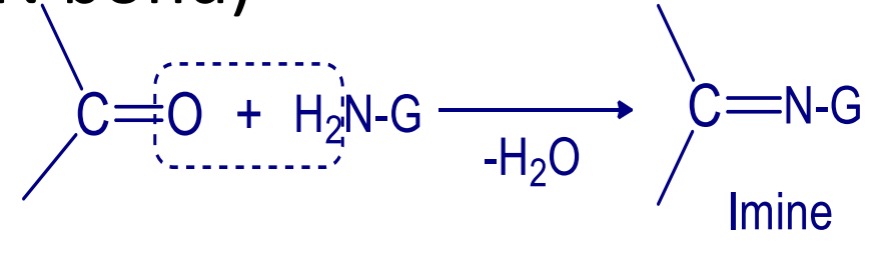
1. Reaction with hydroxyl amine
When aldehyde and ketone reacts with hydroxyl amine then oxime are formed.

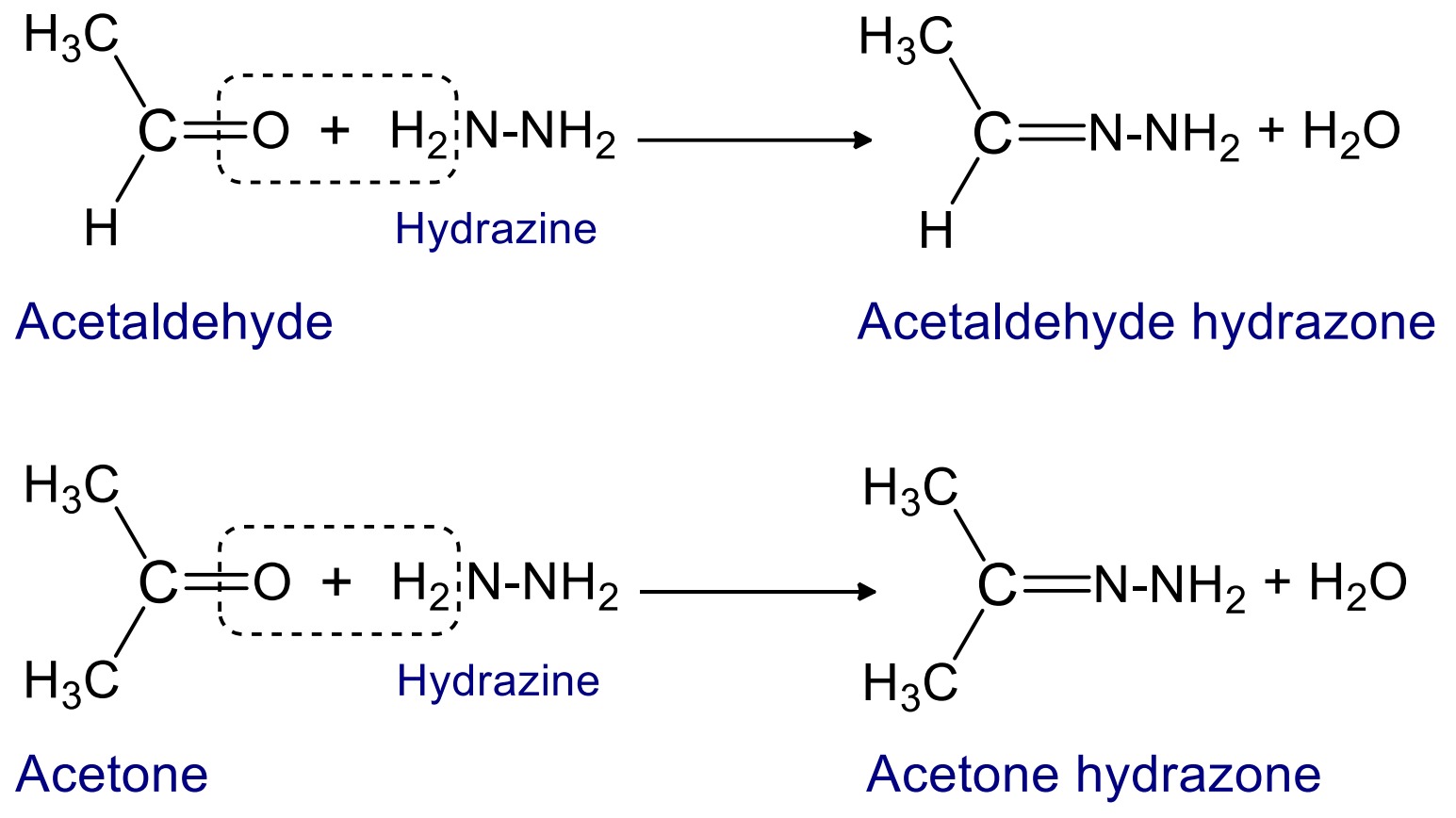
2. Reaction with hydrazine
When aldehyde and ketone reacts with hydrazine then hydrazone are formed.
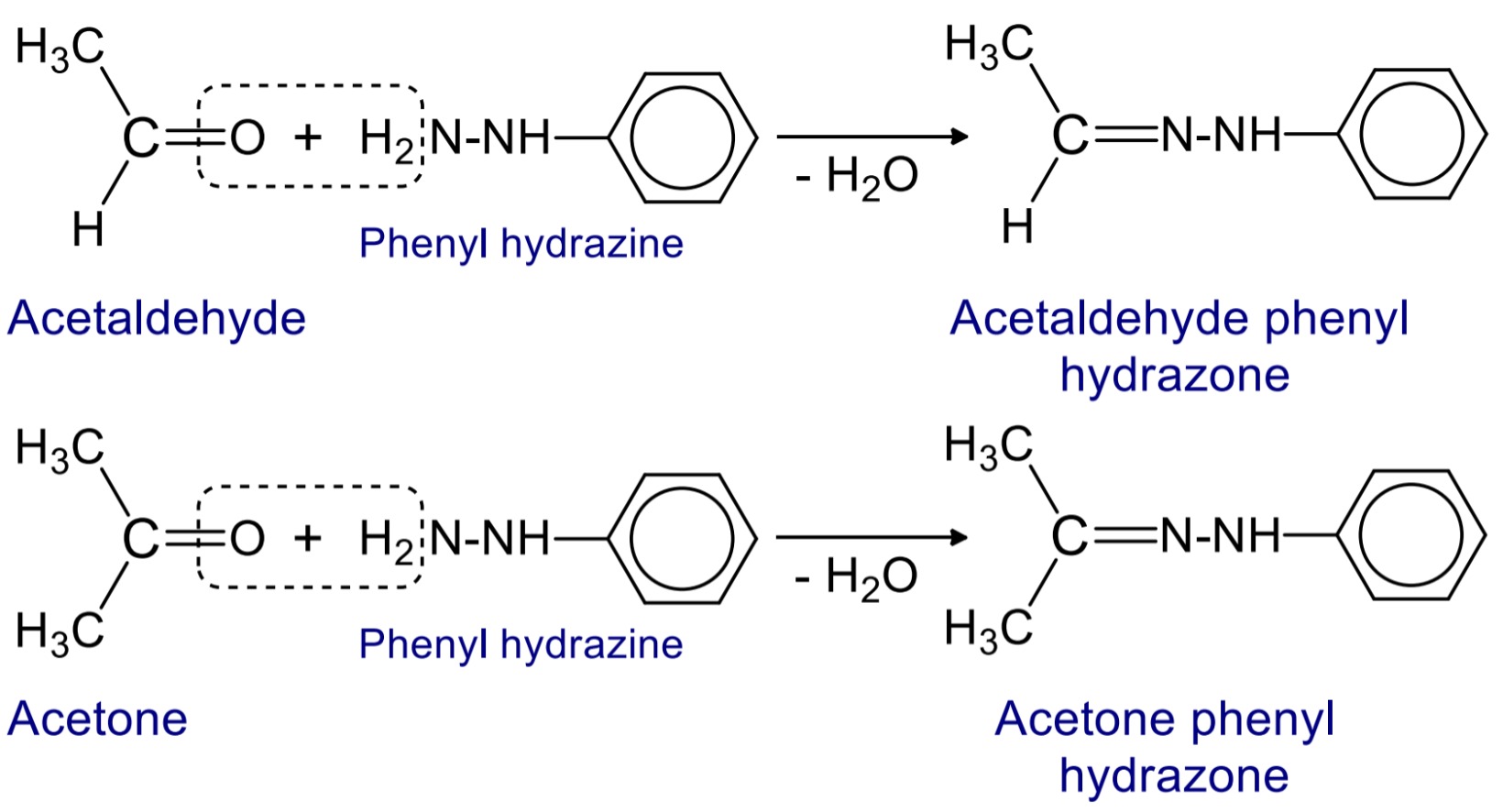
3. Reaction with phenyl hydrazine
When aldehyde and ketone reacts with phenyl hydrazine then phenylhydrazone are formed.
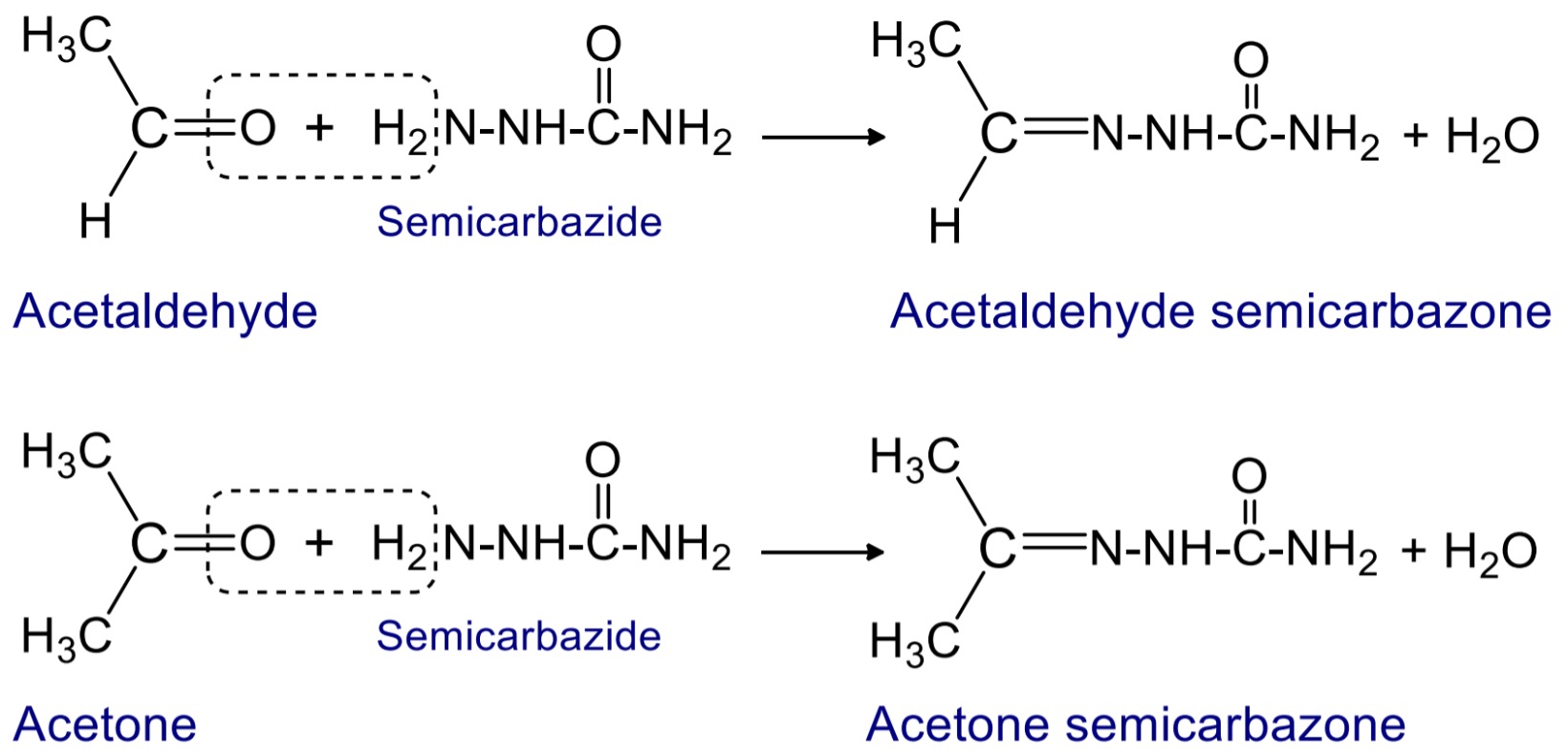
4. Reaction with semicarbazide
When aldehyde and ketone reacts with semicarbazide then crystalline ppt. of semicarbazone is formed.
6. Reaction with 2,4-DNP (2,4-dinitrophenylhydrazine)
When aldehyde and ketone reacts with 2,4-DNP then yellow or orange red crystalline ppt. of 2,4-dinitrophenyl hydrazone is formed. This is the test reaction of aldehyde and ketone.
.jpeg)
[C] Oxidation reaction
1. Oxidation with strong oxidizing agent
When aldehydes are oxidized with strong oxidizing agent then carboxylic acids are formed.

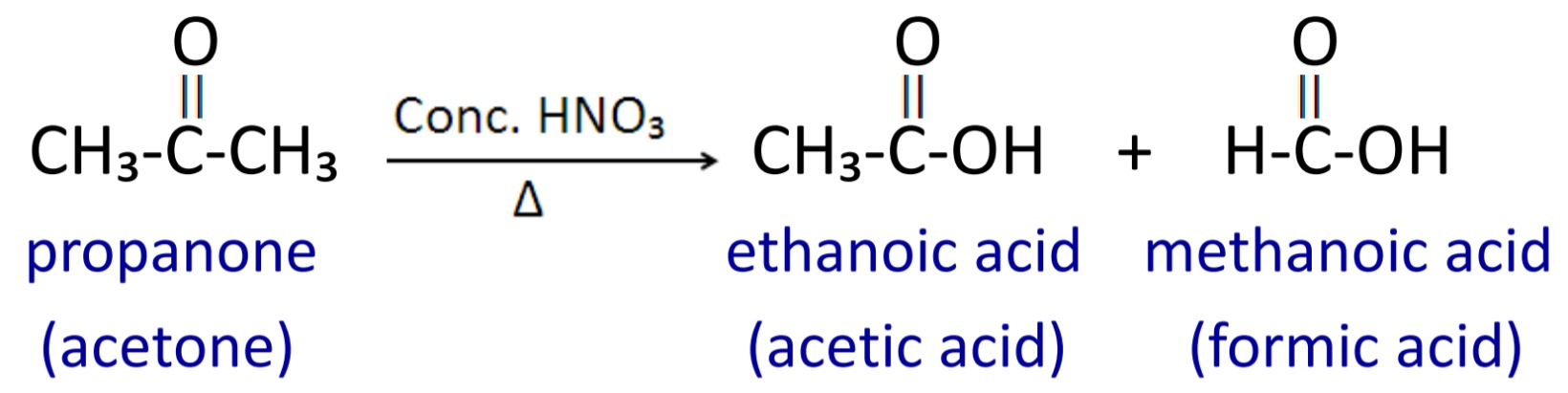
Ketone does not oxidized easily because it has carbon to carbon strong covalent bond. Therefore ketones are oxidized under drastic condition with Conc. Nitric acid at high temperature then carboxylic acid having less number of carbon atom are formed.
2. Oxidation with Tollen’s reagent
An ammoniacal silver nitrate solution is called Tollen’s reagent. When AgNO₃ solution reacts with NaOH followed by NH₄OH then a colourless soluble complex is formed, which is called Tollen’s reagent.
AgNO₃ + NaOH → AgOH↓ + NaNO₃
(grey ppt.)
AgOH + 2NH₄OH → [Ag(NH₃)₂]OH + 2H₂O
Diammine silver(I) hydroxide
(soluble complex) (Tollen’s reagent)
When aldehyde is heated with Tollen’s reagent then the Tollen’s reagent is reduced to metallic silver which is deposited on inner wall of test tube. This reaction is known as silver mirror test or Tollen’s test for aldehyde.

3. Oxidation with Fehling’s solution
An alkaline copper sulphate solution mixed with sodium-potassium tartrate (Rochelle salt) is called Fehling’s solution. When aldehyde is heated with Fehling’s solution then brick red ppt. of cuprous oxide (Cu₂O) is formed.
%20is%20formed.jpeg)
[D] Reduction reaction
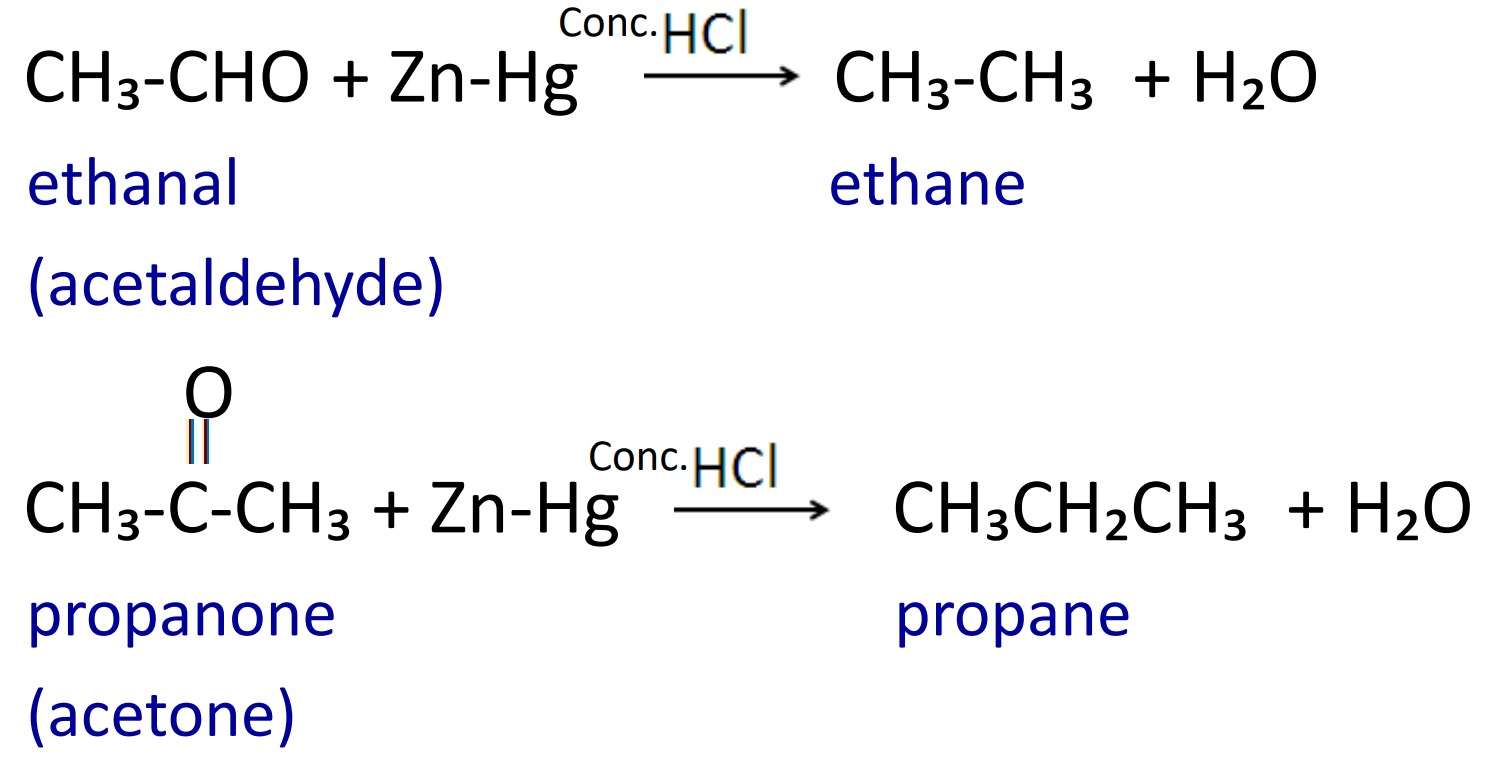
1. Clemmensen reduction
When aldehyde and ketone are reduced with Zn-Hg in presence of conc. HCl then alkane having same number of carbon atom are formed.
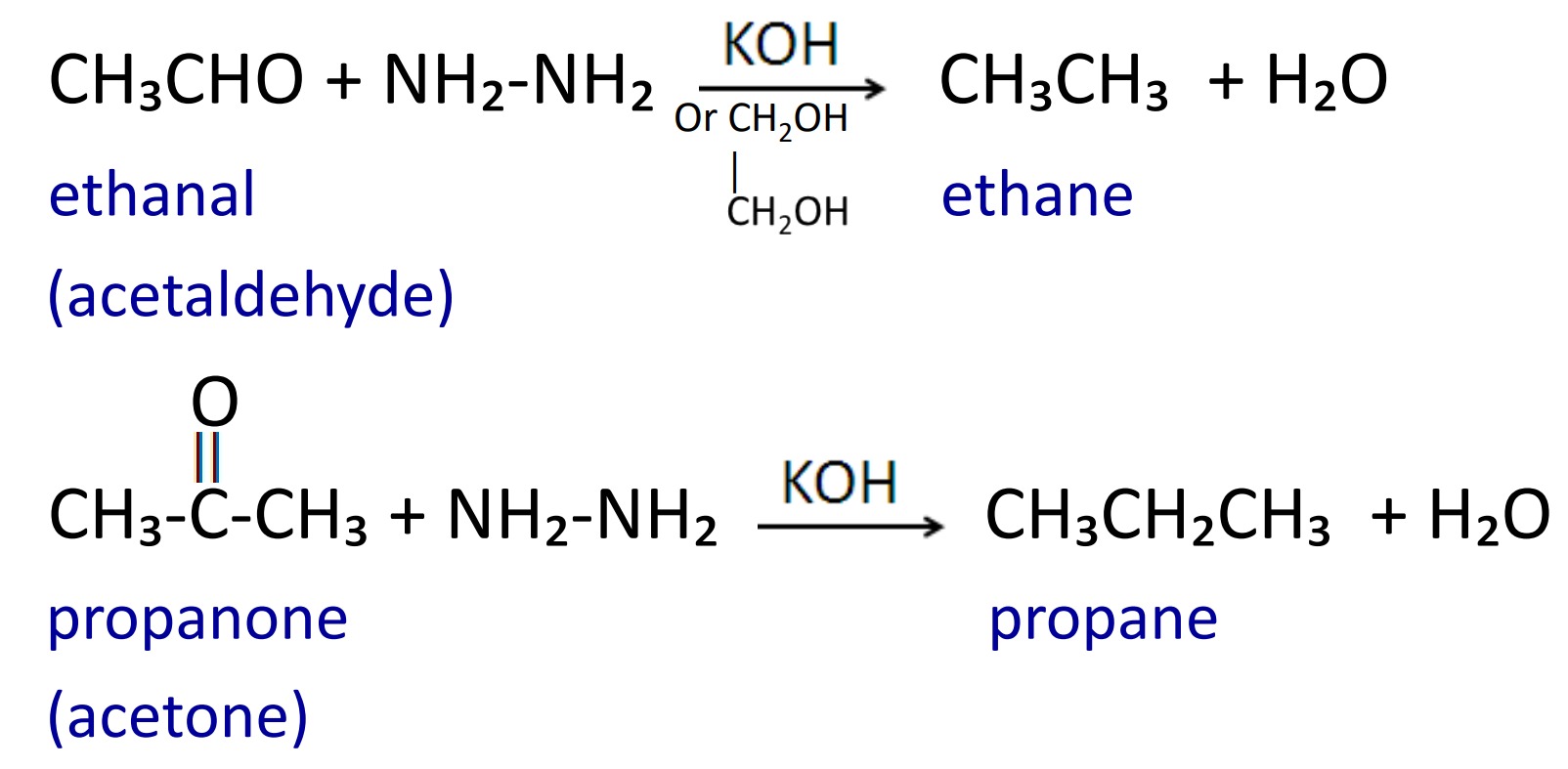
2. Wolff-Kishner reduction
When aldehyde and ketone are reduced with hydrazine in presence of alcoholic KOH or ethylene glycol then alkane having same number of carbon atom are formed, and this reaction is called Wolff-Kishner reduction.
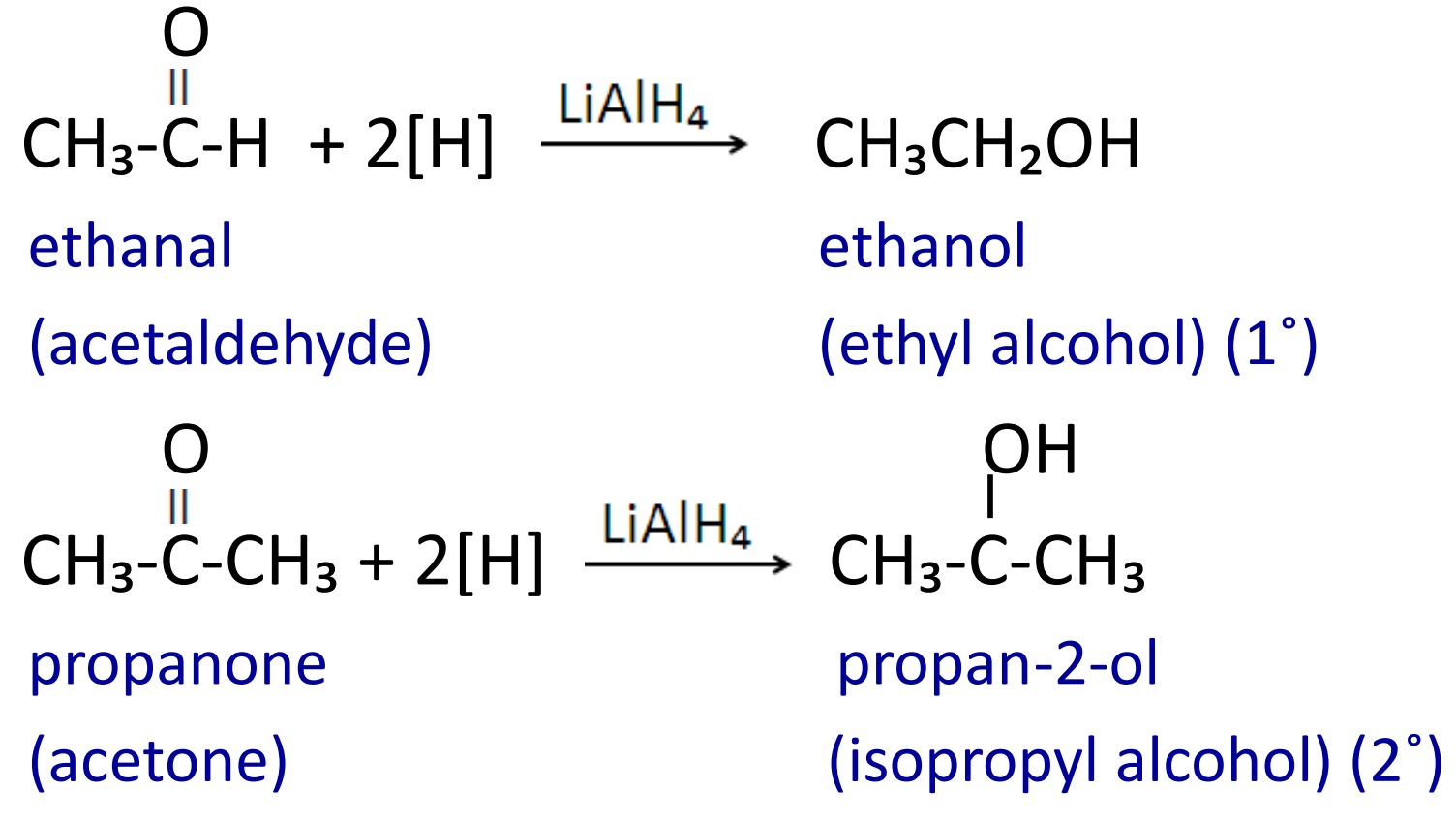
3. Reduction with metal hydride
When aldehyde and ketone are reduced with LiAlH₄ or NaBH₄ then primary and secondary alcohols are formed.
[E] Other reaction
1. Aldol condensation reaction
When aldehydes and ketones having α-hydrogen reacts with dilute NaOH, then they undergo self condensation reaction give β-hydroxy aldehyde or ketone. The compound containing alcohol and carbonyl group is called aldol.

Cross aldol condensation reaction
When two molecules of aldehyde and ketone having alpha hydrogen undergo condensation reaction in presence of dilute NaOH then β-hydroxy aldehyde or ketone are formed.

α−hydrogen of aldehyde is more reactive. So negatively charged carbon of aldehyde attacks to carbonyl carbon of ketone to give β-hydroxy aldehyde.
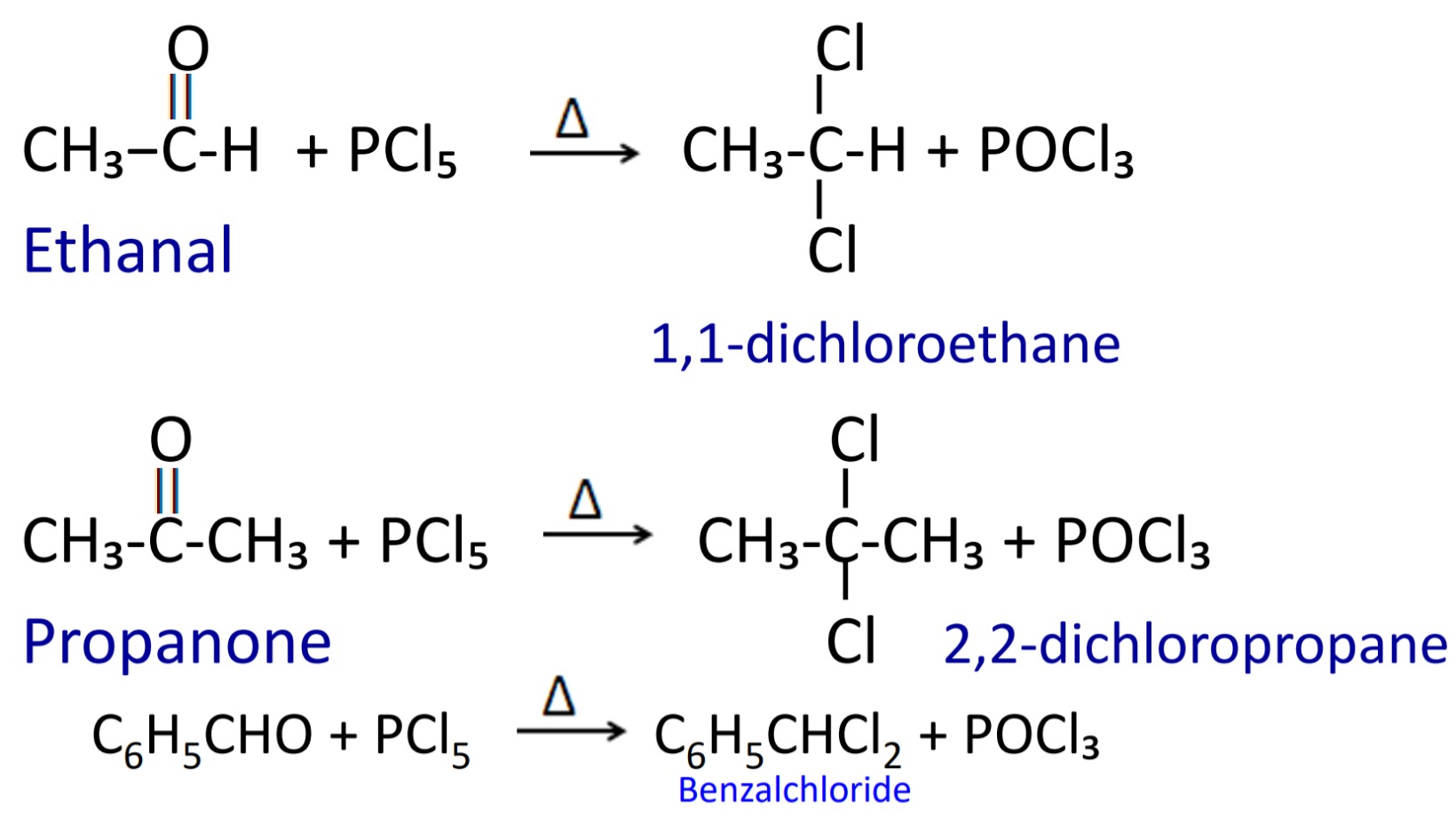
2. Reaction with PCl₅
When aldehyde and ketone reacts with PCl₅ then geminal dichloride are formed.
Special reactions of methanal (formaldehyde)
1. Cannizzaro reaction
When aldehydes having no α-hydrogen undergo self condensation reaction in presence of Conc. NaOH then oxidation and reduction reaction occurs to give sodium salt of acid and alcohol.

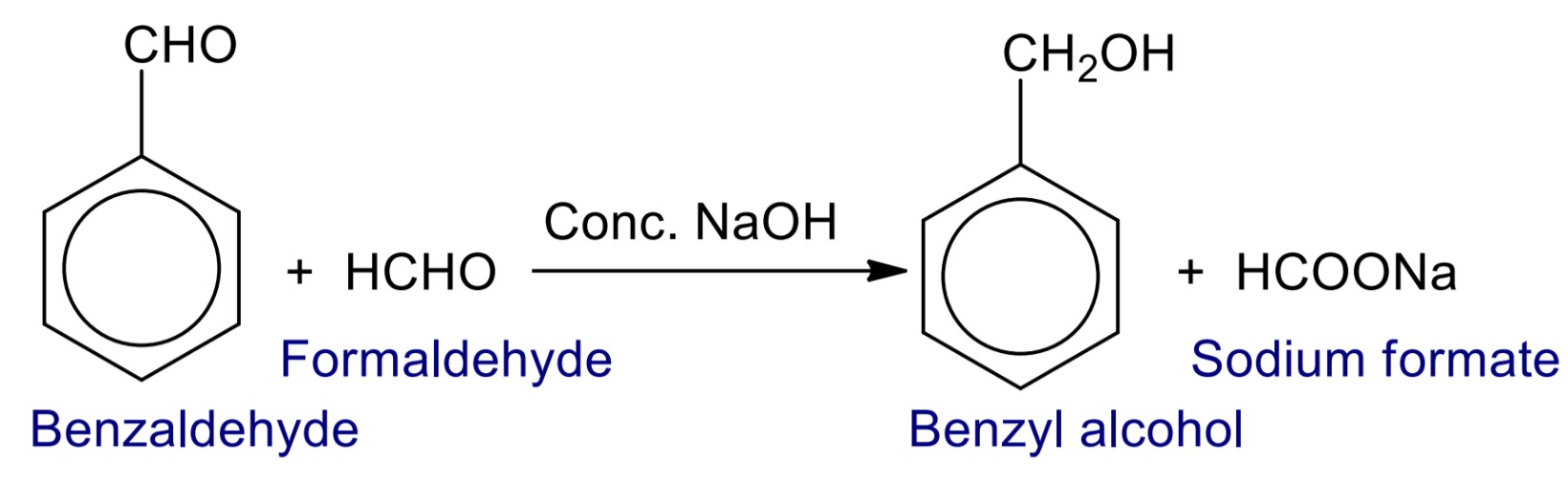
Cross Cannizzaro’s reaction
When two different molecules of aldehyde having no α-hydrogen (formaldehyde and benzaldehyde) undergo self condensation reaction in presence of Conc. NaOH then benzyl alcohol and sodium formate are formed.
2. Reaction with ammonia
When formaldehyde reacts with ammonia then urotropine is formed which is used as anti-septic drug for urinary infection.
6HCHO + 4NH₃ → (CH₂)₆N₄ + 6H₂O
Formaldehyde urotropine
.jpeg) |
| Fig: Urotropine (hexamethylenetetramine) |
3. Reaction with phenol
When formaldehyde reacts with phenol in presence of acid or alkali then a mixture of ortho and para hydroxy benzyl alcohol are formed.

These ortho and para hydroxy benzyl alcohol undergo condensation to give highly cross linked thermosetting polymer of bakelite. (Phenol-formaldehyde resin)

Formalin
40% solution of formaldehyde in water is called Formalin.
Uses of formalin
1. It is used in preservation of biological specimens.
2. It is used in leather industry for silvering of mirrors.
3. Used for manufacture of bakelite, resins etc.
4. Used for synthesis of dyes like pararosaniline and indigo.
5. Used as general antiseptic and preparation of urotropine.




